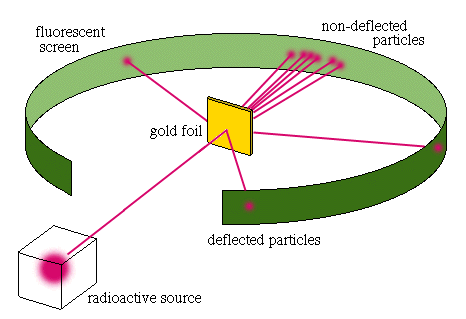
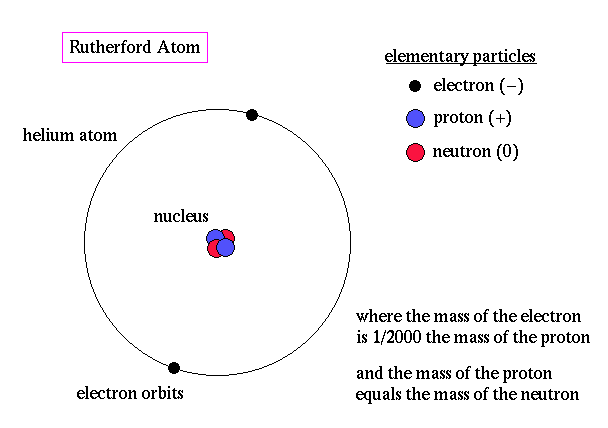
Ernest RutherfordRutherford was a student of Thomson, and continued experimenting atoms. Rutherford shot alpha particles at a thin sheet of gold to observe what happened when the positive passes through the gold atoms. He believed if Thompson’s model was correct that the particles could easily pass through the positive cloud. But they scattered, therefore making Rutherford believe that there was a positively charged nucleus with negative electrons orbiting around
it. |
| History of Atomic Theory |
|